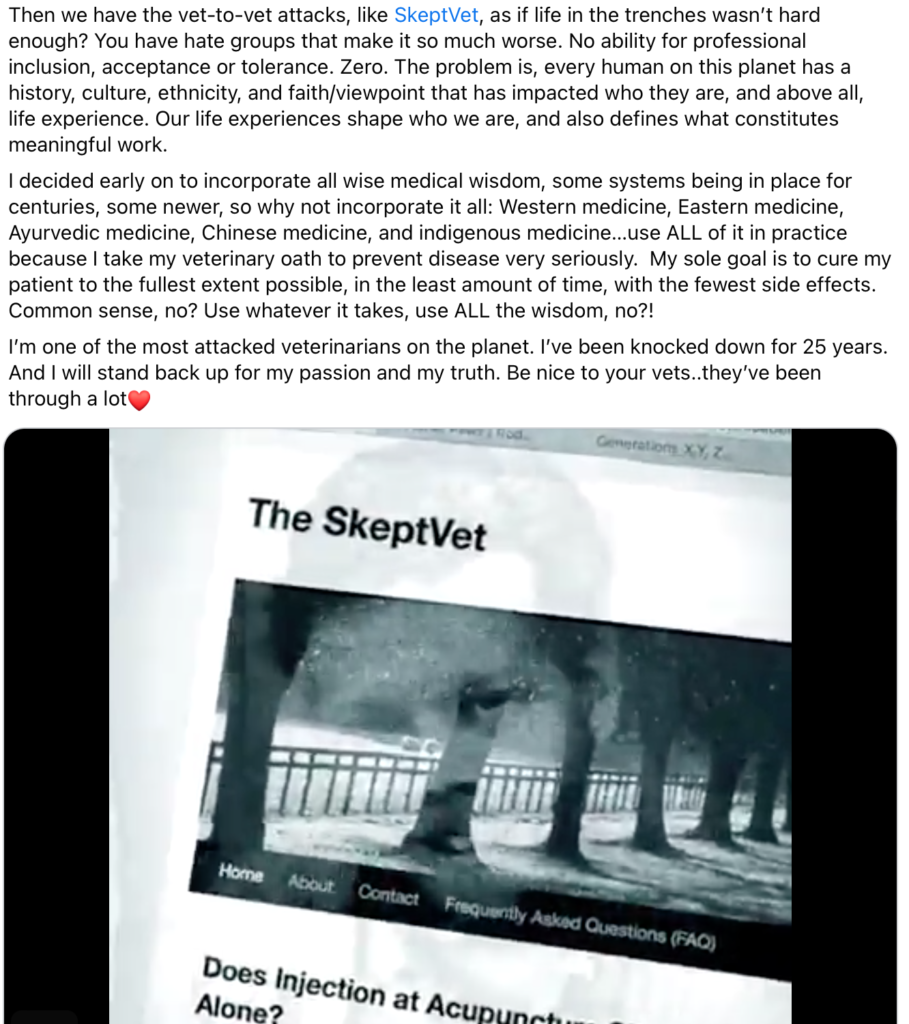
The general trend for life expectancy in humans has been upward for a long time. Improvements in nutrition, sanitation, and both therapeutic and preventative medical care have led to humans today living longer and healthier lives than at any time in history.
The impression most veterinarians have is that the same is true for our pets. Nutrition and healthcare have improved for dogs and cats, as they have for humans, and owned pets who are well cared for are clearly healthier than free-roaming, unowned individuals. However, not nearly as much data is collected on lifespan and mortality patterns in companion animals, so it is difficult to prove that life expectancy is increasing.
I have written about this topic previously here and elsewhere. My conclusions based on the data available then were that it seemed likely dogs, at least, were living longer, but it couldn’t be stated with any confidence. Now, however, there is a bit more data which strengthens the case a bit.
Montoya M, Morrison JA, Arrignon F, Spofford N, Charles H, Hours MA, Biourge V. Life expectancy tables for dogs and cats derived from clinical data. Front Vet Sci. 2023 Feb 21;10:1082102.
This study looked retrospectively (backwards in time) at the huge patient data set collected by the Banfield Pet Hospital group. While there are challenges and limitations in this kind of data set and analysis, it provides a useful source of data for many types of research.
In this case, the authors looked at mortality data and constructed a set of life tables, to get a sense of how long dogs and cats in this population lived and what factors were associated with this. With all of the necessary caveats (discussed in the original paper), the general trend was consistent with most of the previous data; life expectancy seems to be increasing for our pets.
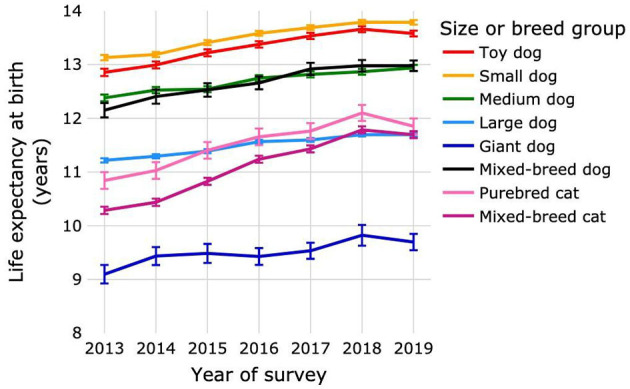
Confidence in this result is increased by the consistency of other findings in the study. For example, overweight dogs tended to have shorter lives, as other studies have shown and as we would expect from much more robust evidence in humans. This relationship, interestingly, was not as clear in cats, and other research has also shown that the relationship between body weight and lifespan in cats is much more complex than typically seen in humans and dogs.
While we are always forced to cope with more limited data in veterinary medicine than our human healthcare colleagues have available, we must make decisions based on the most plausible arguments and highest-quality data we do have. Contemporary nutrition and healthcare for our animal companions are better than in the past, and this appears to be extending their lives as it does ours.
Hysterical claims that our pets are living shorter and more unhealthy lives are based purely on an anti-modernity, anti-science ideology, and they are not consistent with the facts. Examples are easy to find of the dire consequences for rejecting the evidence that modern science have improved health and lifespan; from the huge number of unnecessary deaths form COVID and the resurgence of preventable disease due to vaccine refusal to potentially serious disease associated with the use unconventional diets, this concern about declining health and lifespan is misinformed and can lead to poor healthcare choices for the pets we love.