Aging in the Dog:
Foundations of Canine Geriatric Medicine
What is aging?
How we define aging depends on our goals and our frame of reference. From the biomedical perspective of the veterinary clinician, the important elements are:
- The passage of time
- Deleterious physiologic and functional changes at the molecular, cellular, tissue, and organismal levels
- A progressive increase in the risk of the three Ds
- Disability
- Disease
- Death
As dogs age, they lose robustness (the ability to maintain a state of baseline or optimal physiologic function in the face of external stressors) and resilience (the ability to return to this state following perturbations caused by such stressors).1 This leads to frailty and the development of many age-associated diseases which seem superficially unrelated but which are actually all consequences of the same underlying mechanisms of aging.
Is aging a disease?
Because aging is a universal phenomenon, at least in mammals, and because historically there have been no effective interventions to slow or stop the aging process per se, only treatments to mitigate the clinical consequences, aging is widely seen as natural, inevitable, and immutable. However, decades of foundational research in laboratory model species, and more limited recent studies in humans and companion dogs, suggest that the core mechanisms of aging can be altered in a way that may prevent the health consequences of aging.2,3 Much debate has focused on the semantic issue of whether or not something natural and ubiquitous but also responsible for illness and death should be labeled a disease.4 There is not yet any consensus resolution to this debate.
A pragmatic approach that avoids this semantic debate is to view aging is the most important modifiable risk factor for disease in companion dogs. This is a familiar concept to veterinarians. Obesity, for example, is a risk factor for multiple specific diseases which increases the overall risk of mortality.5 Focusing on reducing this risk by targeting obesity, rather than waiting for the clinical consequences to develop and then managing each independently is a well-established practice in preventative medicine.
Age-associated changes are responsible for most of the health problems of adult dogs, and there are plausible hypotheses suggesting therapies that could directly target aging and so prevent these problems. The focus of geroscience (the study of the fundamental mechanisms of aging) is to identify these targets and therapies so the field of geriatrics (the clinical management of the aged) can move away from the reactive practice of treating the clinical consequences of aging as they arise and towards a preventative approach of delaying and preventing these consequences by modifying the fundamental processes of aging.
Why do dogs age?
It is sometimes supposed that because evolution selects against deleterious traits, the fact that most animals develop disability and disease with age is a paradox. Shouldn’t we have evolved for eternal good health, since this seems obviously more “fit” than getting old and frail?6
One possible explanation for this apparent paradox is that genes and phenotypes which promote reproductive success early in life can cause health problems later in life but still be favored by natural selection.7 Intensive parental investment, for example, may improve reproductive success while simultaneously diminishing the parents reserves and capacity to maintain their own health. Genes favoring such investment would likely outcompete genes favoring parental neglect even if the latter strategy led to longer healthspan and lifespan.
Dogs are also arguably more a product of artificial than natural selection, and many aspects of their aging reflect this. Large and giant-breed dogs, for example, have much shorter lives than smaller breeds, and this is closely tied to genetic differences associated with growth and adult size.8–10
How do dogs age?
The cellular and molecular processes associated with aging, and the tissue dysfunction and ultimate health problems that result from these processes, are complex and multifactorial. Research in laboratory species, and in humans and our canine companions, has elucidated many of these mechanisms, and our understanding of them is growing rapidly.1,11–13 Figure 1 illustrated just a few nodes in the complex web of aging.
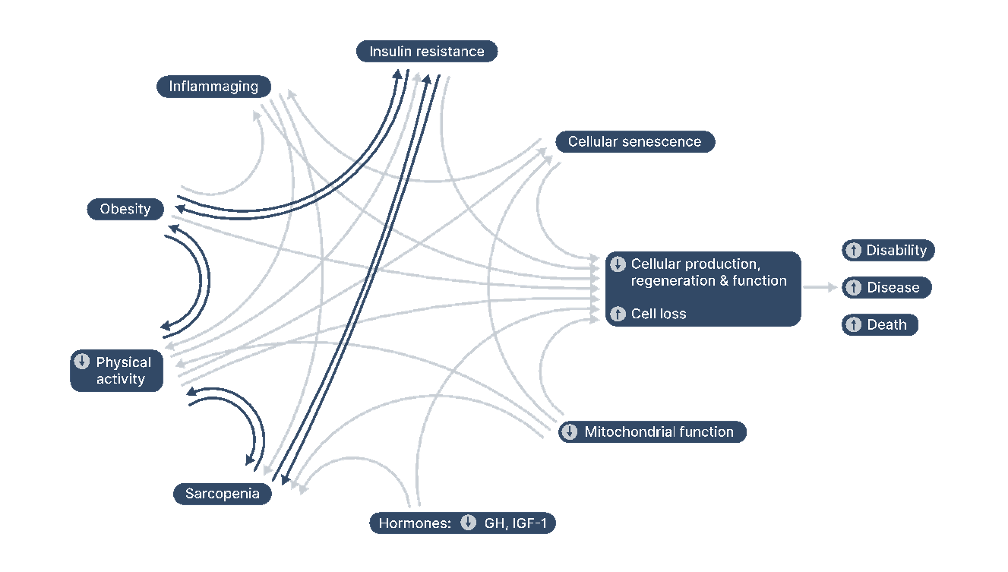
Figure 1. The web of tissue aging—a partial illustration of the interactions between key tissue-aging mechanisms. GH = Growth hormone. IGF-1 = Insulin-like growth factor-1. From McKenzie (2022)1.
Despite the complexity of aging, it is ultimately just biology, a collection of physiologic processes that can be understood and manipulated like any other. There is a vibrant field of canine geroscience research investigating the processes of aging and potential targets for intervention to extend healthspan and lifespan.
When is a dog “old”?
The old canard that every year in a dog’s life is equivalent to seven years for a human is a misleading oversimplification. Dogs age more rapidly than humans at the beginning and end of their life cycles, but the overall lifespan trajectory is quite similar.14 Large and small dogs often age quite differently as well, so the designation of geriatric status may be appropriate much earlier for some breeds than others. In terms of chronological age, or simply the time a dog has been alive, one has to consider size, breed, and individual characteristics. For practical purposes, this approximation is at least useful for triggering more intensive monitoring and investigation of clinical complaints, but it is merely a very rough guide to when we might call a dog “old:”
- Small (under 20 lbs) > 12 years
- Medium (20-50 lbs) > 10 years
- Large (over 50 lbs) > 8 years
More important than chronological age, however, is biological age, defined as the degree to which aging has impacted the robustness, resilience, and state of health and function in an individual as measured by physical, functional, and biomarker assessment.1 We do not yet have reliable tools for measuring biological age, but many are being developed and tested, and ultimately this will be a much more accurate way to predict the age-associated risk of the three Ds (disability, disease, and death) than chronological age.
What can we do about canine aging?
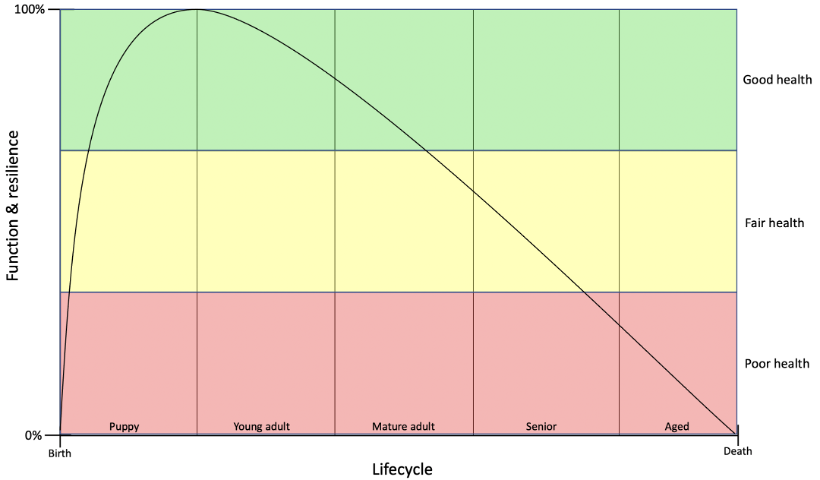
The ideal response to the burden of aging on the health and wellbeing of dogs and their caregivers is to target the core mechanisms of aging and extend lifespan by preventing the entire array of age-associated diseases and clinical problems. Figure 2 illustrates the goal of extending both lifespan (the time alive) and healthspan (the time without significant age-associated health problems).
a)
b)
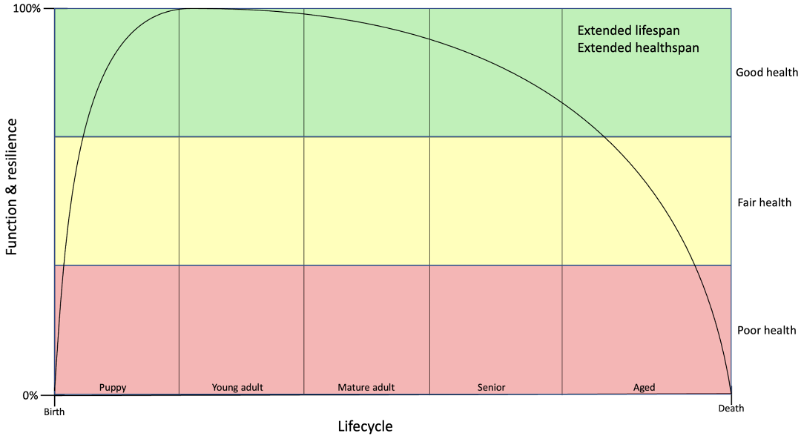
Figure 2. Lifespan trajectories in the dog. a) standard trajectory showing gains and declines in robustness and resilience throughout the lifecycle from birth to death. b) trajectory showing the results of therapies targeting core aging mechanisms and resulting in extended lifespan and healthspan and morbidity compressed into a smaller window of time prior to death
Until we have validated therapies to accomplish this, however, we can best serve our canine patients by encouraging lifestyle habits that are known to delay age-associated disease and mortality:
- Prevent obesity15
- Encourage regular physical activity16
- Encourage consistent, evidence-based veterinary care17
A systematic approach to the geriatric canine patient
Once age-associated health problems do develop, we can best care for our patients with systematic, rational, evidence-based assessment and management. There are many tools that allow us to evaluate pain, impaired mobility, frailty, and other manifestations of canine aging, and these are not yet widely and consistently used. Many of the most common age-associated diseases, such as chronic kidney disease, cardiac disease, and many types of neoplasia, have been the focus of extensive research, and there are often clinical practice guidelines and other evidence-based tools to help support high-quality therapeutic management of these conditions.18–20 And finally, despite some significant limitations, the emerging discipline of hospice and palliative care is an important element in caring for those patients most severely affected by aging.21
A systematic approach to geriatric medicine encourages proactive identification of disease and clinical problems and using the bets available evidence to guide diagnostic and treatment interventions. Too often, clinical signs of frailty and disease are dismissed as “just slowing down” or “normal aging” rather than appropriately assessed, monitored, and managed. In the future, proactive and systematic detection of such signs will be a critical element in the determination of biological age and the decision to employ therapies targeting aging directly.
References
1. McKenzie BA, Chen FL, Gruen ME, Olby NJ. Canine Geriatric Syndrome: A Framework for Advancing Research in Veterinary Geroscience. Front Vet Sci. 2022;0:462. doi:10.3389/FVETS.2022.853743
2. Campisi J, Kapahi P, Lithgow GJ, Melov S, Newman JC, Verdin E. From discoveries in ageing research to therapeutics for healthy ageing. Nat 2019 5717764. 2019;571(7764):183-192. doi:10.1038/s41586-019-1365-2
3. Lawler DF, Evans RH, Larson BT, Spitznagel EL, Ellersieck MR, Kealy RD. Influence of lifetime food restriction on causes, time, and predictors of death in dogs. J Am Vet Med Assoc. 2005;226(2):225-231. doi:10.2460/javma.2005.226.225
4. McKenzie BA. Is Aging a Disease? DVM360. 2022;53(3):25.
5. Salt C, Morris PJ, Wilson D, Lund EM, German AJ. Association between life span and body condition in neutered client-owned dogs. J Vet Intern Med. 2019;33(1):89-99. doi:10.1111/JVIM.15367
6. Johnson AA, Shokhirev MN, Shoshitaishvili B. Revamping the evolutionary theories of aging. Ageing Res Rev. 2019;55:100947. doi:10.1016/J.ARR.2019.100947
7. Austad SN, Hoffman JM. Is antagonistic pleiotropy ubiquitous in aging biology? Evol Med Public Heal. 2018;2018(1):287-294. doi:10.1093/EMPH/EOY033
8. Rimbault M, Beale HC, Schoenebeck JJ, et al. Derived variants at six genes explain nearly half of size reduction in dog breeds. Genome Res. 2013;23(12):1985-1995. doi:10.1101/gr.157339.113
9. Plassais J, Rimbault M, Williams FJ, Davis BW, Schoenebeck JJ, Ostrander EA. Analysis of large versus small dogs reveals three genes on the canine X chromosome associated with body weight, muscling and back fat thickness. Clark LA, ed. PLOS Genet. 2017;13(3):e1006661. doi:10.1371/journal.pgen.1006661
10. Kraus C, Pavard S, Promislow DEL. The size-life span trade-off decomposed: Why large dogs die young. Am Nat. 2013;181(4):492-505. doi:10.1086/669665
11. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194-1217. doi:10.1016/j.cell.2013.05.039
12. Sándor S, Kubinyi E. Genetic Pathways of Aging and Their Relevance in the Dog as a Natural Model of Human Aging. Front Genet. 2019;10:948. doi:10.3389/fgene.2019.00948
13. McKenzie BA. Comparative veterinary geroscience: mechanism of molecular, cellular, and tissue aging in humans, laboratory animal models, and companion dogs and cats. Am J Vet Res. 2022;83(6). doi:10.2460/AJVR.22.02.0027
14. Wang T, Ma J, Hogan AN, et al. Quantitative Translation of Dog-to-Human Aging by Conserved Remodeling of the DNA Methylome. Cell Syst. 2020;11(2):176-185.e6. doi:10.1016/j.cels.2020.06.006
15. Salt C, Morris PJ, Wilson D, Lund EM, German AJ. Association between life span and body condition in neutered client-owned dogs. J Vet Intern Med. 2019;33(1):89-99. doi:10.1111/JVIM.15367
16. McKenzie BA. What does the evidence say about feline fitness and dog aerobics? Vet Pract News. Published online January 2022:25-26.
17. Urfer SR, Wang M, Yang M, Lund EM, Lefebvre SL. Risk Factors Associated with Lifespan in Pet Dogs Evaluated in Primary Care Veterinary Hospitals. J Am Anim Hosp Assoc. 2019;55(3):130-137. doi:10.5326/JAAHA-MS-6763
18. (IRIS) IRIS. Treatment Recommendations for CKD in Dogs.; 2019.
19. Keene BW, Atkins CE, Bonagura JD, et al. ACVIM consensus guidelines for the diagnosis and treatment of myxomatous mitral valve disease in dogs. J Vet Intern Med. 2019;33(3):1127-1140. doi:10.1111/jvim.15488
20. Biller B, Patel M, Smith D, Bryan C. 2016 AAHA Oncology Guidelines for Dogs and Cats*. J Am Anim Hosp Assoc. 2016;52:181-204. doi:10.5326/JAAHA-MS-6570
21. Bishop G, Cooney K, Cox S, et al. 2016 AAHA/IAAHPC End-of-Life Care Guidelines. J Am Anim Hosp Assoc. 2016;52(6):341-356. doi:10.5326/JAAHA-MS-6637)










Fascinating
twitter has celine haloua post and skepvet post saying “next up Skydiving:” google search had nothing for skepvet and Celine combined. watching john stewarts and celines podcast where she is making the claim for a placebo double blind dog study. Trying to find the reason why skepvet and celine post appear together on twitter and a little upset that i am so desperate to live forever that i am trying to get medical information about dog longevity on a comedians podcats.
As I’ve discussed here on the blog a few times, I now work part-time for Loyal, a biotech startup developing therapies to delay age-associated disease and extend lifespan in dogs. Celine in our CEO, and I often repost material related to Loyal and canine aging on various Skeptvet social accounts. We are working towards running field efficacy studies to achieve FDA licensing for some of our therapies, though these have not yet started. As for living forever, not really our goal, and I think folks our age are going to have to stick with the usual lifestyle interventions for now since there are no validated therapeutics to delay aging in humans.
cool,
going to follow Celine on twitter. She mentioned in the podcast a longevity study or proposal in dogs turned in to the FDA. Is that study submitted online somewhere to read? As far as folks living longer now with something besides lifestyle interventions. I would put my money on the new class of wt reduction drugs such as the one Lilly is coming out with. Had to laugh when i saw Celine’s tattoo representing the Labrador longevity study. The girls committed. Reminds me of the old veterinarian whose wife dies after 50 years of marriage and in memory of her death he tattoos her name on his arm.
https://www.cnn.com/2023/01/12/health/reversing-aging-scn-wellness/index.html
seems like a good study to repeat in dogs.
What are your thoughts on rapamycin that has shown increases in healthspan and lifespan across multitudes of mammalian species? I know a good amount of people ( scientists, doctors, lay people, etc) who personally take it and also have been giving it to their cats and dogs. (https://www.rapamycin.news/news) Here is website that talks about people’s experiences on it and giving it to pets.
There is work being done on dogs through the dog aging program, TRIAD (Test of Rapamycin In Ageing Dogs), and the Kaeberlein lab ( https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5411365/). If you have not looked into it, I highly recommend you look into the literature of rapamycin for longevity purposes.
I will attach some papers I have read on the topic:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8190242/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8940555/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6996961/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6814615/
Like so many other drugs, this has encouraging preclinical and animal model evidence for beneficial effects on MTOR-mediated aging-pathways. The DAP pilot study was a precursor to a full clinical trial, which is now in process. I will be very interested in the results of that study. Too early to recommend it for routine use, in my opinion, but wouldn’t surprise me if some kind of rapalog ends up being a useful element to therapies targeting aging, in dogs and humans.
Pingback: Aging in the Dog: Presentation at the Fetch Kansas City Conference - Animal Blog