
]WHAT IS AGING?
How we define aging depends on our goals and our frame of reference. From the biomedical perspective of the veterinary clinician, the important elements are:
- The passage of time
- Deleterious physiologic and functional changes at the molecular, cellular, tissue, and organismal levels
- A progressive increase in the risk of the three Ds
- Disability
- Disease
- Death
As dogs age, they lose robustness (the ability to maintain a state of baseline or optimal physiologic function in the face of external stressors) and resilience (the ability to return to this state
following perturbations caused by such stressors).1 This leads to frailty and the development of many age-associated diseases which seem superficially unrelated but which are actually all consequences of the same underlying mechanisms of aging.
IS AGING A DISEASE?
Because aging is a universal phenomenon, at least in mammals, and because historically there have been no effective interventions to slow or stop the aging process per se, only treatments to mitigate the clinical consequences, aging is widely seen as natural, inevitable, and immutable. However, decades of foundational research in laboratory model species, and more limited recent studies in humans and companion dogs, suggest that the core mechanisms of aging can be altered in a way that may prevent the health consequences of aging.2,3 Much debate has focused on the semantic issue of whether or not something natural and ubiquitous but also responsible for illness and death should be labeled a disease.4 There is not yet any consensus resolution to this debate.
A pragmatic approach that avoids this semantic debate is to view aging is the most important modifiable risk factor for disease in companion dogs. This is a familiar concept to veterinarians. Obesity, for example, is a risk factor for multiple specific diseases which increases the overall risk of mortality.5 Focusing on reducing this risk by targeting obesity, rather than waiting for the clinical consequences to develop and then managing each independently is a well-established practice in preventative medicine.
Age-associated changes are responsible for most of the health problems of adult dogs, and there are plausible hypotheses suggesting therapies that could directly target aging and so prevent these problems. The focus of geroscience (the study of the fundamental mechanisms of aging) is to identify these targets and therapies so the field of geriatrics (the clinical management of the aged) can move away from the reactive practice of treating the clinical consequences of aging as they arise and towards a preventative approach of delaying and preventing these consequences by modifying the fundamental processes of aging.
CANINE GERIATRIC SYNDROME
Regardless of whether one chooses to think of aging as a disease, it is useful to characterize and measure the key elements of canine aging process, including the underlying mechanisms and the clinical phenotype. Canine Geriatric Syndrome (CGS) is both a conceptual framework to organize canine geroscience research and also a potential clinical assessment tool to help determine biological age and predict health outcomes in aging dogs. Figure 1 illustrates the components of CGS.
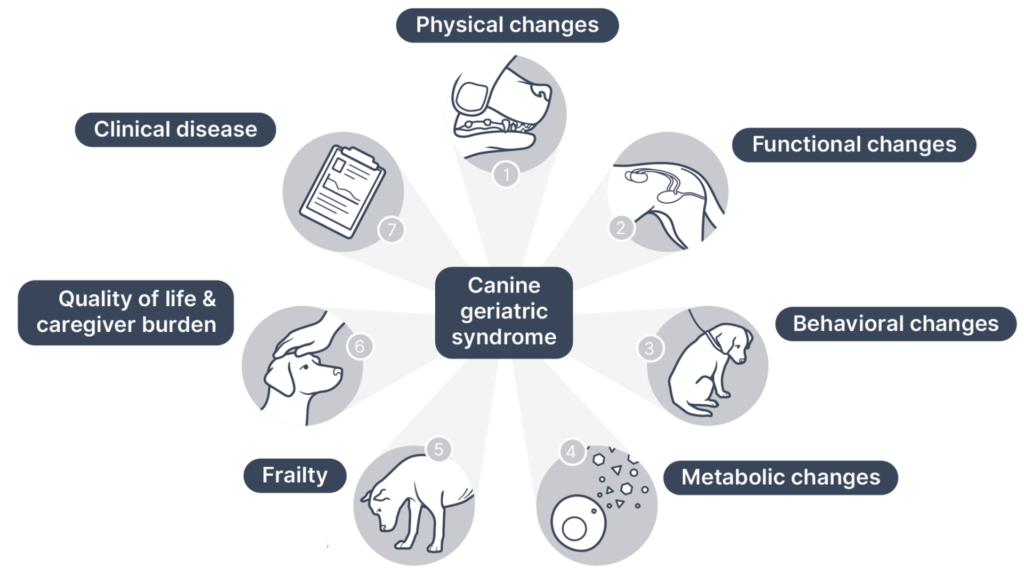
The fundamental mechanisms of aging, from the cellular and molecular level to the level of tissues and organs, generate a phenotype that includes physical, function, behavioral and metabolic changes. These culminate in frailty, which dimmish quality of life, burden human caregivers, and eventually lead to the key negative health outcomes: disability, disease, and death. Currently, this syndrome serves primarily as a scaffold focusing research to better understand the causes and manifestations of canine aging. Eventually, clinical assessment of the syndrome will be possible, leading to a diagnosis of CGS that quantifies the biological age of individuals, predicts morbidity and mortality, and guides clinical intervention.
HOW DO DOGS AGE?
The cellular and molecular processes associated with aging, and the tissue dysfunction and ultimate health problems that result from these processes, are complex and multifactorial. Research in laboratory species, and in humans and our canine companions, has elucidated many of these mechanisms, and our understanding of them is growing rapidly. Typical changes include diminished stem cell number and function, declining mitochondrial number and function, accumulation of aberrant and dysfunctional proteins, and many others.1,6–8
Despite the complexity of aging, it is ultimately just biology, a collection of physiologic processes that can be understood and manipulated like any other. There is a vibrant field of canine geroscience research investigating the processes of aging and potential targets for intervention to extend healthspan and lifespan.
Aging manifests in many visible physical changes, from greying of the coat to lenticular sclerosis and loss of lean body mass. Functional changes include decreased mobility and diminished hearing and vision. Behaviorally, old dogs are often less active, less social, and more prone to anxiety and confusion, with many eventually developing canine cognitive dysfunction, a brain disorder similar in many ways to Alzheimer’s Disease in humans.
WHEN IS A DOG “OLD”?
The old canard that every year in a dog’s life is equivalent to seven years for a human is a misleading oversimplification. Dogs age more rapidly than humans at the beginning and end of their life cycles, but the overall lifespan trajectory is quite similar.9 Large and small dogs often age quite differently as well, so the designation of geriatric status may be appropriate much earlier for some breeds than others. In terms of chronological age, or simply the time a dog has been alive, one has to consider size, breed, and individual characteristics. For practical purposes, this approximation is at least useful for triggering more intensive monitoring and investigation of clinical complaints, but it is merely a very rough guide to when we might call a dog “old:”
- Small (under 20 lbs) > 12 years
- Medium (20-50 lbs) > 10 years
- Large (over 50 lbs) > 8 years
More important than chronological age, however, is biological age, defined as the degree to which aging has impacted the robustness, resilience, and state of health and function in an individual as measured by physical, functional, and biomarker assessment.1 We do not yet have reliable tools for measuring biological age, but many are being developed and tested, and ultimately this will be a much more accurate way to predict the age-associated risk of the three Ds (disability, disease, and death) than chronological age.
AGE-ASSOCIATED DISEASES AND CLINICAL PROBLEMS
Most categories of disease become more common in older dogs, including neoplasia, degenerative musculoskeletal diseases (e.g. osteoarthritis and sarcopenia), cardiac disease (e.g. myxomatous mitral valve disease and cardiomyopathy), chronic kidney disease, and others. Apart from congenital disorders, infections, and trauma, most of the significant diseases and causes of death dogs experience result from the progressive accumulation of cellular and tissue changes associated with aging.
Important clinical problems often develop which are not always associated with a specific diagnosis of disease. Loss of mobility, pain, sensory and cognitive decline, and many other such aging changes diminish quality of life, increase caregiver burden, and eventually lead to euthanasia. The common view of these as “natural” and inevitable part of the lifecycle impedes appropriate assessment and management of these conditions and delays necessary research to develop preventative interventions to extend healthspan.
A SYSTEMATIC APPROACH TO ASSESSMENT & MANAGEMENT OF GERIATRIC DOGS
Once age-associated health problems do develop, we can best care for our patients with systematic, rational, evidence-based assessment and management. There are many tools that allow us to evaluate pain, impaired mobility, frailty, and other manifestations of canine aging, and these are not yet widely and consistently used. Many of the most common age-associated diseases, such as chronic kidney disease, cardiac disease, and many types of neoplasia, have been the focus of extensive research, and there are often clinical practice guidelines and other evidence-based tools to help support high-quality therapeutic management of these conditions.13–15 And finally, despite some significant limitations, the emerging discipline of hospice and palliative care is an important element in caring for those patients most severely affected by aging.16
ASSESSMENT AND MANAGEMENT TOOLS
An important part of a systematic approach to geriatric dogs is consistent, objective assessment and management of age-related diseases and clinical problems. This is best accomplished with the regular use of validated clinical metrology instruments and reliance, when possible, on evidence-based clinical practice guidelines. There are many CMIs available to quantify pain, mobility, frailty, quality of life, and the burden of care on the owners of geriatric dogs. Repeated use of these allows detection of changes over time, better evaluation of the effectiveness of therapies than unaided observation, and better communication with owners to facilitate decision making.
There are also numerous clinical practice guidelines available to support management of the most common age-associated problems and diseases. While some are more rigorous and evidence-based than others, all offer rational and systematic approaches that can help clinicians make comprehensive treatment and management plans.
PITFALLS IN CANINE GERIATRICS
A systematic approach to geriatric medicine encourages proactive identification of disease and clinical problems and using the best available evidence to guide diagnostic and treatment interventions. Too often, clinical signs of frailty and disease are dismissed as “just slowing down” or “normal aging” rather than appropriately assessed, monitored, and managed. In the future, proactive and systematic detection of such signs will be a critical element in the determination of biological age and the decision to employ therapies targeting aging directly.
References
1. McKenzie BA, Chen FL, Gruen ME, Olby NJ. Canine Geriatric Syndrome: A Framework for Advancing Research in Veterinary Geroscience. Front Vet Sci. 2022;0:462. doi:10.3389/FVETS.2022.853743
2. Campisi J, Kapahi P, Lithgow GJ, Melov S, Newman JC, Verdin E. From discoveries in ageing research to therapeutics for healthy ageing. Nat 2019 5717764. 2019;571(7764):183-192. doi:10.1038/s41586-019-1365-2
3. Lawler DF, Evans RH, Larson BT, Spitznagel EL, Ellersieck MR, Kealy RD. Influence of lifetime food restriction on causes, time, and predictors of death in dogs. J Am Vet Med Assoc. 2005;226(2):225-231. doi:10.2460/javma.2005.226.225
4. McKenzie BA. Is Aging a Disease? DVM360. 2022;53(3):25.
5. Salt C, Morris PJ, Wilson D, Lund EM, German AJ. Association between life span and body condition in neutered client-owned dogs. J Vet Intern Med. 2019;33(1):89-99. doi:10.1111/JVIM.15367
6. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013;153(6):1194-1217. doi:10.1016/j.cell.2013.05.039
7. Sándor S, Kubinyi E. Genetic Pathways of Aging and Their Relevance in the Dog as a Natural Model of Human Aging. Front Genet. 2019;10:948. doi:10.3389/fgene.2019.00948
8. McKenzie BA. Comparative veterinary geroscience: mechanism of molecular, cellular, and tissue aging in humans, laboratory animal models, and companion dogs and cats. Am J Vet Res. 2022;83(6). doi:10.2460/AJVR.22.02.0027
9. Wang T, Ma J, Hogan AN, et al. Quantitative Translation of Dog-to-Human Aging by Conserved Remodeling of the DNA Methylome. Cell Syst. 2020;11(2):176-185.e6. doi:10.1016/j.cels.2020.06.006
10. Salt C, Morris PJ, Wilson D, Lund EM, German AJ. Association between life span and body condition in neutered client-owned dogs. J Vet Intern Med. 2019;33(1):89-99. doi:10.1111/JVIM.15367
11. McKenzie BA. What does the evidence say about feline fitness and dog aerobics? Vet Pract News. Published online January 2022:25-26.
12. Urfer SR, Wang M, Yang M, Lund EM, Lefebvre SL. Risk Factors Associated with Lifespan in Pet Dogs Evaluated in Primary Care Veterinary Hospitals. J Am Anim Hosp Assoc. 2019;55(3):130-137. doi:10.5326/JAAHA-MS-6763
13. (IRIS) IRIS. Treatment Recommendations for CKD in Dogs.; 2019.
14. Keene BW, Atkins CE, Bonagura JD, et al. ACVIM consensus guidelines for the diagnosis and treatment of myxomatous mitral valve disease in dogs. J Vet Intern Med. 2019;33(3):1127-1140. doi:10.1111/jvim.15488
15. Biller B, Patel M, Smith D, Bryan C. 2016 AAHA Oncology Guidelines for Dogs and Cats*. J Am Anim Hosp Assoc. 2016;52:181-204. doi:10.5326/JAAHA-MS-6570
16. Bishop G, Cooney K, Cox S, et al. 2016 AAHA/IAAHPC End-of-Life Care Guidelines. J Am Anim Hosp Assoc. 2016;52(6):341-356. doi:10.5326/JAAHA-MS-6637)
SUMMARY INCLUDING 5 KEY “TAKE HOME” POINTS
- Aging is not an inevitable and immutable fact of life. It is a modifiable risk factor for the most causes of disability, disease, and death in adult dogs.
- Canine Geriatric Syndrome describes the physical, function, behavioral, and metabolic changes that occur in aging dogs that lead to clinical disease, frailty, diminished quality of life, and a significant caregiver burden on dog owners.
- Aging is just biology! The cellular, molecular, and tissue changes that occur, and that lead to Canine Geriatric Syndrome, are complex but comprehensible. The better we understand these changes, the better we can intervene to mitigate them.
- A systematic, evidence-based approach to assessment and management of diseases and clinical problems in geriatric dogs, supported by validated clinical metrology instruments and clinical practice guidelines, is a more efficient, comprehensive, and effective strategy for high-quality patient care.









